
There has been a longstanding discussion regarding the oversight and regulation of laboratory developed tests (LDTs). The Clinical Laboratory Improvement Amendments (CLIA), which was enacted by Congress in 1988, governs the accreditation, inspection, and certification process for laboratories. Under CLIA, the Centers for Medicare & Medicaid Services (CMS) has oversight of clinical laboratories and LDTs (note that Washington State and New York State have their own laboratory standards and labs are required to receive a valid permit from the state itself). The United States Food and Drug Administration (FDA) states that the Medical Device Amendments Act of 1976 provides the agency with the authority to regulate LDTs to ensure safety, effectiveness, and design and manufacture quality. However, the FDA has used “enforcement discretion”, allowing laboratories to use LDTs that have not been approved or cleared by the FDA. The FDA, however, has continuously voiced to reconsider its role in regulating LDTs, as evidenced by the FDA’s related draft guidance in 2014.
The focus of LDT regulatory authority is now the topic of two opposing bills in Congress. The Verifying Accurate Leading-Edge IVCT Development (VALID) Act of 2021 would authorize FDA to regulate in vitro clinical tests (IVCTs), unifying oversight over LDTs and in vitro diagnostics (IVDs). In contrast, the Verified Innovative Testing in American Laboratories (VITAL) Act of 2021 would strengthen CLIA and empower CMS to continue to regulate LDTs.
There are countless publications and opinions on the VALID or VITAL act. For example, the American Association for Clinical Chemistry (AACC) has published multiple articles on the topic. To understand the debate, let’s take a refresher on all the requirements and steps a laboratory must undertake to implement and offer an LDT under the current regulations.
High-level overview of LDT regulations and requirements
This high-level overview is not a comprehensive summary of regulations and requirements for diagnostics / medical devices. Its sole intention is to provide a basic understanding of the current regulatory environment. Also, it is important to note that this is an evolving process with ongoing changes.
What is an LDT under the current regulation?
The FDA and CMS both agree that an LDT is “a type of in vitro diagnostic test that is designed, manufactured, and used within a single laboratory.” As discussed above, LDTs are currently governed through CLIA and regulated by CMS while FDA exercises “enforcement discretion.” Any in vitro diagnostic test that does not conform to the definition above is considered an IVD medical device, and subject to FDA regulations and oversight.
Many laboratories develop LDTs out of necessity and market need. LDTs often cover areas, such as rare diseases, that lack commercially available IVDs. LDTs are often the fastest option to respond to new developments and needs. It is also important to mention that, under current laws and regulations, any modification to the approved workflow of an IVD device will render the test as an LDT. In other words, if a laboratory is performing an IVD assay using a non-IVD-approved instrument, the assay will be considered an LDT.
LDTs can utilize a wide variety of applications with different complexities, ranging from molecular diagnostics to immunoassays and mass spectrometry. It must be pointed out that by default - independent of the design - all LDTs fall into the CLIA “high complexity” test category. Tests can be designed to detect a single biomarker or multiple analytes, targeting small molecules, proteins, cells, DNA or RNA. Assays can be developed to identify a wide spectrum of targets, such as pathogens, pharmacogenomic and companion diagnostic markers, oncology markers, genes or proteins linked to rare diseases, or the presence/absence of specific antibodies.
What is the path from development to launch for an LDT under the current regulation?
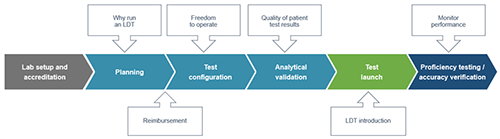
Licensure and certification
Clinical laboratories performing LDTs must be certified under CLIA (unless exempt) and licensed by the states they operate in. The approval criteria for LDTs may vary between states, with New York State having the most stringent review requirements. To receive payment from CMS (or most private payers), clinical laboratories must comply with CLIA and state requirements.
Quality management and documentation
A quality management system (QMS) is a requirement for all CLIA-certified laboratories. The QMS specifies the processes, procedures, responsibilities, data management, and continuous improvement required for achieving quality objectives. The QMS ensures that all product specifications, including equipment and reagents, all personnel trainings, qualifications, and competencies are recorded and updated as required.
From development to implementation of an LDT
One of the first steps to plan the development of an LDT is to define the testing needs, followed by an assessment of commercially available IVDs. If no appropriate IVD test is available, the laboratory needs to assess technical feasibility and draft a development plan to define the intended use of the test and the assay design. A freedom to operate analysis should be performed to understand the intellectual property landscape and identify potential patent infringement issues. It is equally important to review and plan reimbursement strategies early on to ensure the efforts and costs of LDT development will be worthwhile.
The laboratory must develop and optimize the testing procedure. During the development phase, reagents, controls, consumables, and calibrators are specified. Detailed standard operating procedures must be created and approved. The performance characteristics relating to analytical validity must be established according to CLIA and state requirements. Guidelines facilitating analytical validations are published by Clinical Laboratory Standards Institute (CLSI) or International Organization for Standardization (ISO).
Similar to analytical validation study requirements for IVDs, analysis for LDTs include accuracy, precision, analytical sensitivity, analytical specificity, reportable range, reference interval, and other appropriate performance characteristics. In contrast to IVDs, however, analytical validation studies for LDTs are laboratory-specific and limited to the specific conditions of the testing laboratory. Overall, the performance characteristics of the LDT describe the quality of patient test results and is reviewed by CMS and its accreditation organizations. Another major difference between IVD and LDT validation requirements is that the FDA does require clinical validation which is not specifically addressed under CLIA. It is important to note that demonstrating clinical utility of an LDT may be required for reimbursement purposes.
Proficiency testing and accuracy verification requirements
Assessing a test’s performance does not end with the development of an LDT. CMS has published a list of all tests that require proficiency testing (PT). PT involves testing of unknown samples in the same manner as that of patient samples. PT is required for IVDs and LDTs alike and must be performed regularly (usually at least three times a year). CMS and its accreditation organizations review PT results to monitor laboratory performance.
If PT is not required for the type of test offered (such as a CLIA-waived IVD test, or a test type that is not included on the list published by CMS), accuracy verification must still be conducted at least twice a year.
Table 1. Comparison between IVD and LDT requirements
|
Description |
IVD |
LDT |
|
State registration of laboratory |
Yes |
Yes |
|
CLIA certification category |
Depending on IVD complexity |
High complexity |
|
QMS required |
Yes |
Yes |
|
Analytical Validation |
Yes (Performed by manufacturers and reviewed by FDA) |
Yes (Specific to developing laboratory, reviewed by CMS) |
|
Clinical Validation |
Yes |
No |
|
Reimbursement codes |
Usually provided by manufacturers |
Laboratory is responsible (Laboratory often demonstrate clinical utility andbenefits of test for diagnosis, prognosis or treatment decision) |
|
FDA reviewed |
Yes |
Enforcement discretion |
|
Proficiency testing/ accuracy verification |
Yes |
Yes |
How to learn more about current LDT regulation
While the longstanding discussions on LDT oversight continue, it is helpful to understand current regulations and requirements for LDTs, and its differences in comparison to IVDs (Table 1). To further support our readers, we’ve compiled a series of blog articles that cover various aspects of LDT development and implementation:
- The simple, sensible & still spell binding seven questions about laboratory developed tests
- Challenges of establishing laboratory developed tests
- 7 things to know before establishing a laboratory developed test
- Regulatory guidance for clinical laboratories
- LDT and IVD reimbursement in the U.S. explained
Industry Insights articles are created and paid for by advertisers. The views expressed in these articles do not necessarily represent AACC’s views, and their inclusion in CLN is not an endorsement by CLN or AACC.

