In this short article I would like to point out some issues about HbA1c testing that may not be clear in the literature. These are issues related to terminology, standardization, point of care (POC) HbA1c methods and HbA1c test interferences.
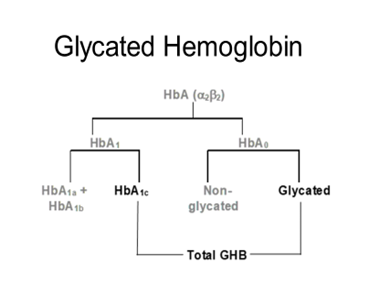
Terminology - GHB, A1C, A1 often confused: Glycated hemoglobin (GHB) is the attachment of glucose to hemoglobin at several different Hb sites. HbA1c (also called A1C) is a specific glycated hemoglobin where glucose is attached to the N-terminal beta chain of hemoglobin. The original Electrophoresis and HPLC methods separated HbA, based on charge, into HbA1 and HbA0 (1) (figure 1). HbA1 was subsequently further separated into HbA1a, HbA1b, HbA1c, HbA1d, etc. The HbA1c peak was shown to be related to diabetes (2) and was later found to be glucose bound to Hb specifically at the N-terminal valine. HbA1 is not the same as total GHB; HbA1 does not include HbA0, while total glycated hemoglobin includes all glycated hemoglobins including glycated HbA0 (hemoglobin glycated at sites other than the beta chain terminus). Since HbA1c was the specific analyte measured in the Diabetes Control and Complications Trial (3) and the United Kingdom Prospective Diabetes Study (4), all clinical recommendations derived from these trials are provided in %HbA1c units. This is the reason for standardizing all GHB assays to report %HbA1c. Some methods measure total GHB while others measure HbA1c specifically; but through the standardization process, they can all report results as %HbA1c.
IFCC and NGSP Standardization compared: At the time of implementation of the NGSP in 1996, there was no higher order reference method or reference material available for HbA1c. In 2004, an IFCC Working Group on HbA1c developed a Reference System for HbA1c (5). The IFCC reference measurement procedure for HbA1c is calibrated with reference materials consisting of pure HbA1c and HbA0 and quantifies the glycated and nonglycated hexapeptides by mass spectrometry or by capillary electrophoresis. HbA1c is thus defined and measured based on its molecular structure and is reported in SI units – mmol/mol. This reference method is then used to assign values to secondary reference materials that can be used to transfer accuracy to routine methods. Both the NGSP and IFCC reference measurement procedures are listed on the JCTLM database as “higher order to a manufacturer’s method”, but the IFCC method is considered a higher order method than the NGSP Reference method. Although there was an excellent linear correlation (r>0.99) between results of the two networks (NGSP and IFCC), the values are not the same. Since all clinical trial data and diabetes recommendations are based on NGSP/DCCT/UKPDS HbA1c values, many countries (including the US) have elected to report HbA1c in % units. By contrast, some countries (predominantly in Europe) have chosen to report results only in IFCC mmol/mol. A few countries are reporting results in both units. Since comparability of the two systems is essential for worldwide use, a master equation was developed between results of the two networks, to allow patient results to easily be converted from one system to the other. This relationship is monitored by twice-yearly comparisons to insure consistency in the relationship over time. The NGSP and IFCC approaches to the standardization of HbA1c results serve different, but complementary purposes. The primary purpose of the IFCC program is to ensure that manufacturers are traceable to an accuracy base of highest order. The NGSP defines acceptable limits for method performance that are based on clinical need, i.e., recommendations for diabetes care by clinical societies.
Point of Care (POC) HbA1c: In the US, accredited clinical laboratories must participate in proficiency testing for all testing performed. However, POC tests that are waived by the Clinical Laboratory Improvement Amendments (CLIA) under a specific regulation are not required to participate in PT. In addition, no expertise is required for personnel who perform waived testing. There are many POC HbA1c tests that have been approved for use in the U.S., and they have also been given waived status. The NGSP certifies many POC HbA1c tests, but NGSP certification is only performed once each year under ideal circumstances by the manufacturer. There is very limited proficiency testing data from waived POC settings and thus there is no way to see how well these tests perform in the clinical setting. This is the main argument against use of POC HbA1c for diagnosis of diabetes where accuracy is extremely important. In addition, the American Diabetes Association Guidelines state that “In the absence of unequivocal hyperglycemia, diagnosis requires two abnormal test results from the same sample or in two separate test samples.” But, often, clinicians rely on one test result. If the one test result is from a waived HbA1c POC test, there is a possibility of misdiagnosis.
HbA1c Test interference: There are two types of interferences with HbA1c test results. First, there are biological interferences. HbA1c measurements depend on both blood glucose concentration and the lifespan of the erythrocyte. Any condition that alters the lifespan of the erythrocytes can affect the interpretation of HbA1c test results. These conditions include hemolytic anemia, Iron iron deficiency anemia, Sickle sickle cell or HbC disease and others. In these cases, other tests such as glycated albumin or fructosamine should be used to assess glycemic status.
The most common cause of analytical interference is hemoglobin variants; these variant interferences are method-specific. The most common hemoglobin variants are Hbs S, C, D and E. In the homozygous form (e.g., HbSS or Sickle cell anemia) these may cause changes in erythrocyte lifespan. However, when present in the heterozygous form (e.g., HbS trait), HbA1c can generally be measured reliably with an appropriate assay. One or more of these variant traits, HbAS, HbAC, HbAE, HbAD as well as elevated HbF (>15%) can interfere with some HbA1c methods. The NGSP regularly evaluates HbA1c methods that are commonly used in the US to determine the degree of interference from these variants. Results of these studies (and those from other authors) are summarized and posted on the NGSP web site (6). Most current methods are not affected by HbF <15% and some methods can measure HbA1c in the presence of HbF as high as 30%. While some methods can detect the presence of Hb variants (e.g., ion-exchange HPLC and capillary electrophoresis methods), others (e.g., immunoassays, boronate affinity and enzymatic methods) cannot.
Unfortunately, only a small number of POC methods have been evaluated for interference from Hb variants and it is not possible to discern if a Hb variant is present when using a POC method since these are usually boronate affinity, immunoassay or enzymatic methods. Therefore, even if this interference is stated in the package insert, the user would not know if the variant is present in the sample and an inaccurate result may be reported.
The bottom line is that for the vast majority of patients with diabetes, HbA1c is a useful indication of mean glycemia and risks for diabetes complications. Clinicians need to work closely with their laboratory if there is a discrepancy between the HbA1c result and either the patient’s glucose results or clinical impression.
References
- AG Schnek and WA Schroeder. The relation between the minor components of whole normal human adult hemoglobin as isolated by chromatography and starch block electrophoresis. J Am Chem Soc 1961;83:1472-8
- Rahbar S; Blumenfeld O; Ranney HM (1969). "Studies of an unusual hemoglobin in patients with diabetes mellitus". Biochem. Biophys. Res. Commun. 1969;36:838–43.
- The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329:977-86.
- U.K. Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998;352:837-53.
- Jeppsson J-O, Kobold U, Barr J, Finke A, Hoelzel W, Hoshino T, Miedema K, Mauri P, Mosca A, Paroni R, Thienpont L, Umemoto M, Weykamp CW. Approved IFCC reference method for the measurement of HbA1c in human blood. Clin Chem Lab Med 2002; 40: 78-89
- NGSP. HbA1c Assay Interferences. https://ngsp.org/interf.asp . (Accessed July 2023).